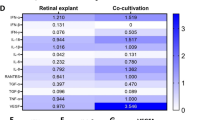
Summary
At the back of the eye, the outermost cell layer of the retina, the pigmented epithelium, lies against a basement membrane that is adjacent to the choroidal vessels that supply the outer sensory retina. During pathogenesis, these interfaces become damaged, and the homeostatic balance between the retinal pigment epithelium (RPE) and the choroidal vessels becomes disrupted, leading to choroidal neovascularization and blindness. To study the cell interactions at the back of the eye, we have used a coculture system in which a stable RPE monolayer has been cultured on a transwell insert and placed over a collagen gel sandwich into which choroidal endothelial cells (CECs) have been seeded. RPE cells have been stimulated by an inflammatory cytokine, interleukin-1 (IL-1β), and the ability of the underlying choroidal endothelium to form vascular tubes has been tested. IL-1β stimulation of the RPE insert increased the number of tubes formed by CECs in the gel as early as 3 d. By 7 d, tubes began to regress. Both IL-8 and monocyte chemotactic protein-1 (MCP-1) were found to be secreted in greater amounts in stimulated RPE. Because MCP-1 is also a chemokine for monocytes, which in turn secrete angiogenic factors, monocytes were added to the upper surface of the choroidal gel sandwich and then incubated with the stimulated RPE insert as above. By day 7, more tubes formed and there was no regression over the experimental time period. The versatility of this model has been illustrated in that both RPE and CECs can be cultured in a more natural construct and their molecular interactions tested by physiologically altering one cell type and not the other.
Similar content being viewed by others
References
Adams, D. H.; Lioyd, A. R. Chemokines: leukocyte recruitment and activation cytokines. Lancet 349:490–495; 1997.
Bikfalvi, A.; Cramer, E. M.; Tenza, D.; Tobelem, G. Phenotypic modulations of human umbilical vein endothelial cells and human dermal fibroblasts using two angiogenic assays. Biol. Cell 72:275–278; 1991.
Bruno, V.; Vittel, D.; Feige, J.-J. In vitro models of vasculogenesis and angiogenesis. Lab. Invest. 81:439–452; 2001.
Cai, J.; Nelson, K. C.; Wu, M.; Sternberg, P.; Jones, D. P. Oxidative damage and protection of the RPE. Prog. Retin. Eye Res. 19(2):205–221; 2000.
Campochiaro, P. A. Cytokine production by retinal pigmented epithelial cells. Int. Rev. Cytol. 146:75–82; 1993.
Campochiaro, P. A. Retinal and choroidal neovascularization. J. Cell. Physiol. 184:301–310; 2000.
Campochiaro, P. A.; Hackett, S. F.; Vinores, S. A. Growth factors in the retina and retinal pigment epithelium. Prog. Retin. Eye Res. 15:547–567; 1996.
Campochiaro, P. A.; Soloway, P.; Ryan, S. J.; Miller, J. W. The pathogenesis of choroidal neovascularization in patients with age-related macular degeneration. Mol. Vis. 5:34; 1999.
Crane, I. J.; Kuppner, M. C.; McKillop-Smith, S.; Knott, R. M.; Forrester, J. V. Cytokine regulation of RANTES production by human retinal pigment epithelial cells. Cell Immunol. 184:37–44; 1998.
Crane, I. J.; Kuppner, M. C.; McKillop-Smith, S.; Wallace, C. A.; Forrester, J. V. Cytokine regulation of granulocyte-macrophage colony-stimulating factor (GM-CSF) production by human retinal pigment epithelial cells. Clin. Exp. Immunol. 115:288–293; 1999.
de, B. J.; Hack, C. E.; Verhoeven, A. J., et al. Chemoattractant and neutrophil degranulation activities related to interleukin-8 in vitreous fluid in uveitis and vitreoretinal disorders. Invest. Ophthalmol. Vis. Sci. 34:3376–3385; 1993.
Dunn, K. C.; Aotaki-Keen, A. E.; Putkey, F. R.; Hjelmeland, L. M. ARPE-19, a human retinal pigment epithelial cell line with differentiated properties. Exp. Eye Res. 62:155–169; 1996.
Elner, S. G.; Elner, V. M.; Jaffe, G. J.; Stuart, A.; Kunkel, S. L.; Strieter, R. M. Cytokines in proliferative diabetic retinopathy and proliferative vitreoretinopathy. Curr. Eye Res. 14:1045–1053; 1995.
Fuentes, M. E.; Durham, S. K.; Swerdel, M. R.; Lewin, A. C.; Barton, D. S.; Megill, J. R.; Bravo, R.; Lira, S. A. Controlled recruitment of monocyte and macrophages to specific organs through transgenic expression of monocyte chemoattractant protein-1. J. Immunol. 155:5769–5776; 1995.
Goede, V.; Brogelli, L.; Ziche, M.; Augustin, H. G. Induction of inflammatory angiogenesis by monocyte chemoattractant protein-1. Int. J. Cancer 82:765–770; 1999.
Green, W. R.; Enger, C. Age-related macular degeneration histopathologic studies. Ophthalmology 100:1519–1535; 1993.
Grossniklaus, H. E.; Cingle, K. A.; Yoon, Y. D.; Ketkar, N.; L'Hernault, N.; Brown, S. Correlation of histologic 2-dimensional reconstruction and confocal scanning laser microscopic imaging of choroidal neovascularization in eyes with age-related maculopathy. Arch. Ophthalmol. 118:625–629; 2000.
Grunwald, J.; Hariprasad, S.; DuPont, J.; Maguire, M.; Fine, S.; Brucker, A.; Maguire, A.; Ho, A. Foveolar choroidal blood flow in age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 39:385–390; 1998.
Holtkamp, G. M.; De Vos, A. F.; Peek, R.; Kijlsta, A. Analysis of the secretion pattern of monocyte chemotactic protein-1 (MCP-1) and transforming growth factor-beta 2 (TGF-β2) by human retinal pigment epithelial cells. Clin. Exp. Immunol. 118:35–40; 1999.
Holtkamp, G. M.; Van Rossem, M.; De Vos, A. F.; Willekens, B.; Peck, R.; Kijlsta, A. Polarized secretion of IL-1 and IL-8 by human retinal pigment epithelial cells. Clin. Exp. Immunol. 112:34–43; 1998.
King, G.; Suzuma, K. Pigment-epithelium-derived factor—a key coordinator of retinal neuronal and vascular functions. N. Engl. J. Med. 342:349–351; 2000.
Ment, L. R.; Stewart, W. B.; Scaramuzzino, D.; Madri, J. A. An in vitro three-dimensional coculture model of cerebral microvascular angiogenesis and differentiation. In Vitro Cell. Dev. Biol. 33A:684–691; 1997.
Montesano, R.; Orci, L.; Vassalli, J. D. In vitro rapid organization of endothelial cells into capillary-like networks is promoted by collagen matrices. J. Cell. Biol. 97(5):1648–1652; 1983 [part 1].
Mousa, S. A.; Lorelli, W.; Campochiaro, P. A. Role of hypoxia and extracellular matrix-integrin binding in the modulation of angiogenic growth factors secretion by retinal pigment epithelial cells. J. Cell Biochem. 74:135–143; 1999.
Murata, T.; He, S.; Hangai, M., et al.. Peroxisome proliferator-activated receptor-γ ligands inhibit choroidal neovascularization. Invest. Ophthalmol. Vis. Sci. 41:2309–2317; 2000.
Murata, T.; Yoshikawa, H.; Hata, Y.; Tsutsumi, C., et al. The role of monocyte chemoattractant protein-1 expression in the pathogenesis of choroidal neovascularization. Invest. Ophthalmol. Vis. Sci. 42:S226; 2001.
Nicosia, R. F.; Ottinetti, A. Modulation of microvascular growth and morphogenesis by reconstituted basement membrane gel in three-dimensional cultures of rat aorta: a comparative study of angiogenesis in matrigel, collagen, fibrin, and plasma clot. In Vitro Cell. Dev. Biol. 26:119–128; 1990.
Oh, H.; Takagi, H.; Takagi, C., et al. The potential angiogenic role of macrophages in the formation of choroidal neovascular membranes. Invest. Ophthalmol. Vis. Sci. 40:1891–1898; 1999.
Sakamoto, T.; Sakamoto, H.; Hinton, D. R.; Spee, C.; Ishibashi, T.; Ryan, S. J. In vitro studies of human choroidal endothelial cells. Curr. Eye Res. 14:621–627; 1995a.
Sakamoto, T.; Sakamoto, H.; Murphy, T. L.; Spee, C.; Soriano, D.; Ishibashi, T.; Hinton, D. R.; Ryan, S. J. Vessel formation by choroidal endothelial cells in vitro is modulated by retinal pigment epithelial cells. Arch. Ophthalmol. 113:512–520; 1995b.
Sakamoto, T.; Spee, C.; Scuric, Z.; Gordon, E. M.; Hinton, D. R.; Anderson, W. F.; Ryan, S. J. Ability of retroviral transduction to modify the angiogenic characteristics of RPE cells. Graefes Arch. Clin. Exp. Ophthalmol. 236:220–229; 1998.
Salcedo, R.; Ponce, M.; Young, H. A.; Wasserman, K.; Ward, J. M.; Kleinman, H. K.; Oppenheim, J. J.; Murphy, W. J. Human endothelial cells express CCR2 and respond to MCP-1: direct role of MCP-1 in angiogenesis and tumor progression. Blood 96:34–40; 2000.
Scherberich, A.; Beretz, A. Culture of vascular cells in tridimensional (3−D) collagen: a methodological review. Therapie 55:35–41; 2000.
Stephan, H.; Spee, C.; Murata, T.; Cui, J. Z.; Ryan, S. J.; Hinton, D. R. Rapid isolation of choriocapillary endothelial cells by lycopersicon esculentum-coated Dynabeads. Graefes Arch. Clin. Exp. Ophthalmol. 236:779–784; 1998.
Strieter, R. M.; Kunkel, S. L.; Elner, V. M., et al. Interleukin-8: a corneal factor that induces neovascularization. Am. J. Pathol. 141:1279–1284; 1992.
Yang, S.; Graham, J.; Kahn, J. W.; Schwartz, E. A.; Gerritsen, M. E. Functional role for PECAM-1 (CD31) and VE-cadherin (CD144) in tube assembly and lumen formation in three-dimensional collagen gels. Am. J. Pathol. 155:887–895; 1999.
Yoshida, A.; Yoshida, S.; Khalil, A. K.; Ishibashi, T.; Inomata, H. Role of NF-κB-mediated interleukin-8 expression in intraocular neovascularization. Invest. Ophthalmol. Vis. Sci. 39:1097–1106; 1998.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fan, W., Zheng, J.J. & McLaughlin, B.J. An in vitro model of the back of the eye for studying retinal pigment epithelial-choroidal endothelial interactions. In Vitro Cell.Dev.Biol.-Animal 38, 228–234 (2002). https://doi.org/10.1290/1071-2690(2002)038<0228:AIVMOT>2.0.CO;2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1290/1071-2690(2002)038<0228:AIVMOT>2.0.CO;2